010. Fitting Multi-Component Kinetics using Compunetics I
By Daniel Davies on December 14, 2020
Metoprolol is a cardioselective beta-blocker commonly used for the treatment of hypertension, for which kinetic information would help in the process development stage of manufacture at any pharmaceutical company. Metoprolol is formed from the epoxide opening of the starting material (SM) by isopropylamine, however, metoprolol can then perform a consecutive reaction to form the undesired di-product. The reaction data[1] was obtained from a flow reactor platform and inputted in the Experimentation Toolbox of Compunetics to be fitted.
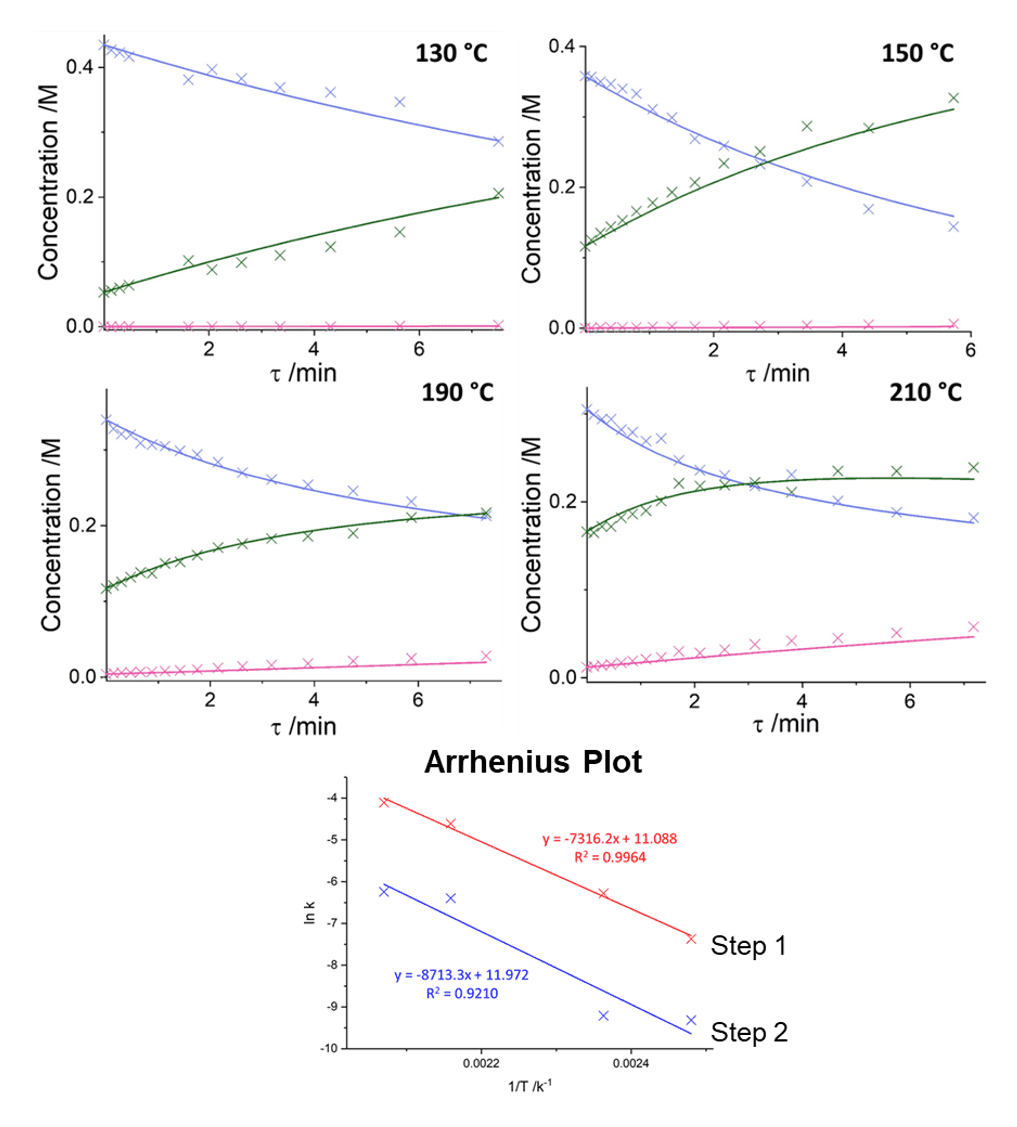
Four experiments were run at differing concentrations and temperatures (130 - 210 °C), then each experiment was fitted individually using the kinetic model:
Using Compunetics to quickly and easily fit these kinetics means that a good relationship can be found for the temperature dependence of the reaction rate, as shown in the Arrhenius plot. As a result, the physical model is shown to be accurate and can be used for further simulations or process optimisation. The kinetic parameters for the formation of metoprolol were determined to be: k190 °C = 9.95 x 10-3 ± 0.07 x 10-3 M-1 s-1, Ea = 60.8 ± 2.0 kJ mol-1 and for the formation of di-product: k190 °C = 1.67 x 10-3 ± 0.40 x 10-3 M-1 s-1, Ea = 72.4 ± 1.9 kJ mol-1.
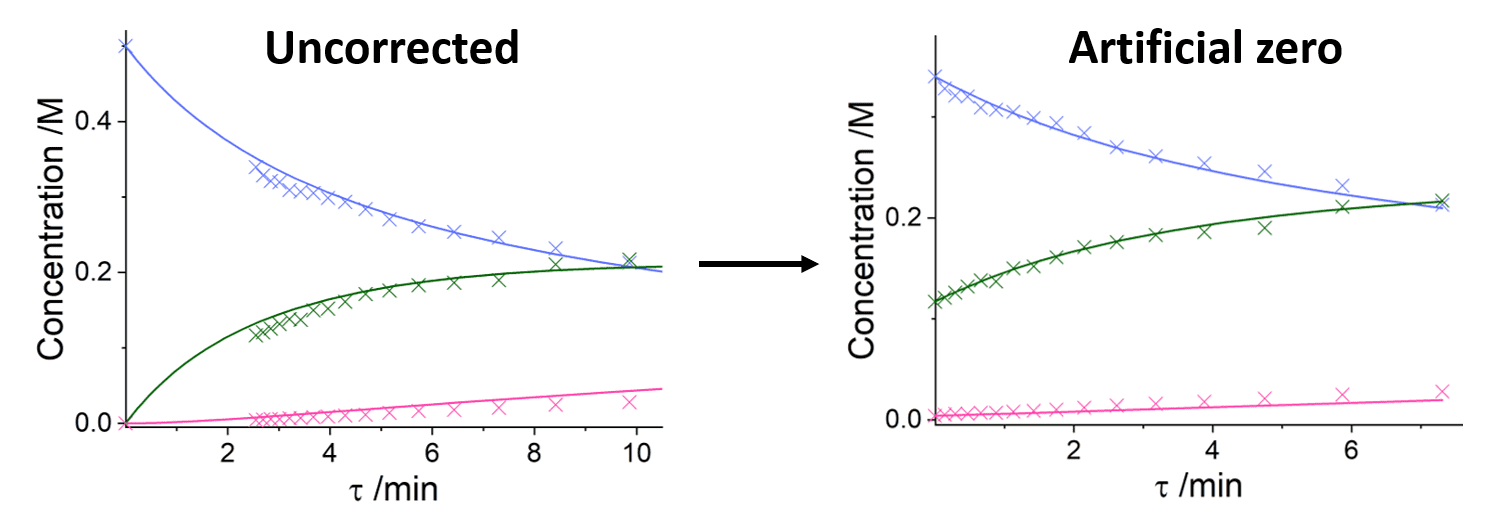
It can also be noted that each kinetic plot has been fitted with an 'artificial zero', meaning that at the experimental time zero there is already reaction conversion. This technique of fitting kinetics with an artificial zero is akin to Variable Time Normalisation Analysis (VTNA) methodologies[2] where the time axis is shifted, thereby asserting an experimental 'start time' when concentrations are known. This can fix any fitting discrepancies that may occur as the first measured time point may not be accurate - it is assumed however, that all time points following this first measurement are correct with respect to the first measurement. It is likely that using these assumptions give a greater accuracy when conducting kinetic fitting. In the case of this experiment, small deviations in asserting the time of the initial measurement may occur from thermal expansion of the solvent because of the very high temperatures, or from pump flow rate errors. This can be seen more clearly below in a comparison of 'uncorrected' and 'artificial zero' fitting at 190 °C, as the kinetic curve already does not fit at the first data point:
Using this acquired kinetic information for process optimisation in Compunetics is then easy using the Simulation Toolbox and/or the machine-learning based optimiser in the Investigator Toolbox. This late-stage reaction in the manufacture of metoprolol can be fully understood from the utilisation of kinetic analysis to build a physical model using Compunetics. The easy-to-use Experimentation Toolbox in Compunetics quickly provides quantitative analysis of any chemical system, allowing total process understanding without the need for high-level coding or mathematical expertise.
- Taylor, C. J. et al., Rapid Automated Determination of Reaction Models and Kinetic Parameters, Chemical Engineering Journal, 2020, 127017.
- Burés, J. Variable time normalization analysis: General graphical elucidation of reaction orders from concentration profiles. Angewandte Chemie, 2016, 128, 52, 16318-16321.