009. Reaction Simulation for Side-Product Prediction II
By Alice Payne on December 07, 2020
One of the major benefits of conducting kinetic analysis is that a reaction can be simulated at different starting concentrations and temperatures, providing that the kinetic parameters are known. These simulations can be very important as reaction outcome predictions can be obtained with confidence without running laboratory experiments, therefore saving time and money to conduct. We recently showed the impact of an organic chemist conducting simulations in Compunetics for the synthesis of paracetamol and why sub-optimal yields are often achieved, all without any expertise in coding or mathematics.
If we consider the same paracetamol synthesis from 4-aminophenol and acetic anhydride, we can expect at some point in the reaction to observe diacetamate formation from Step Two as a byproduct. As we showed in a previous post, an undergraduate experimenter rarely achieves greater than a 70 % yield of paracetamol, likely due to degradation of the acetic anhydride to unreactive acetic acid. However, it is often claimed that a researcher must be careful in this synthesis as not to accidentally obtain impure paracetamol due to the formation of the over-reacted diacetamate. But this begs the question, how much byproduct is actually formed? Is any diacetamate is formed at all?
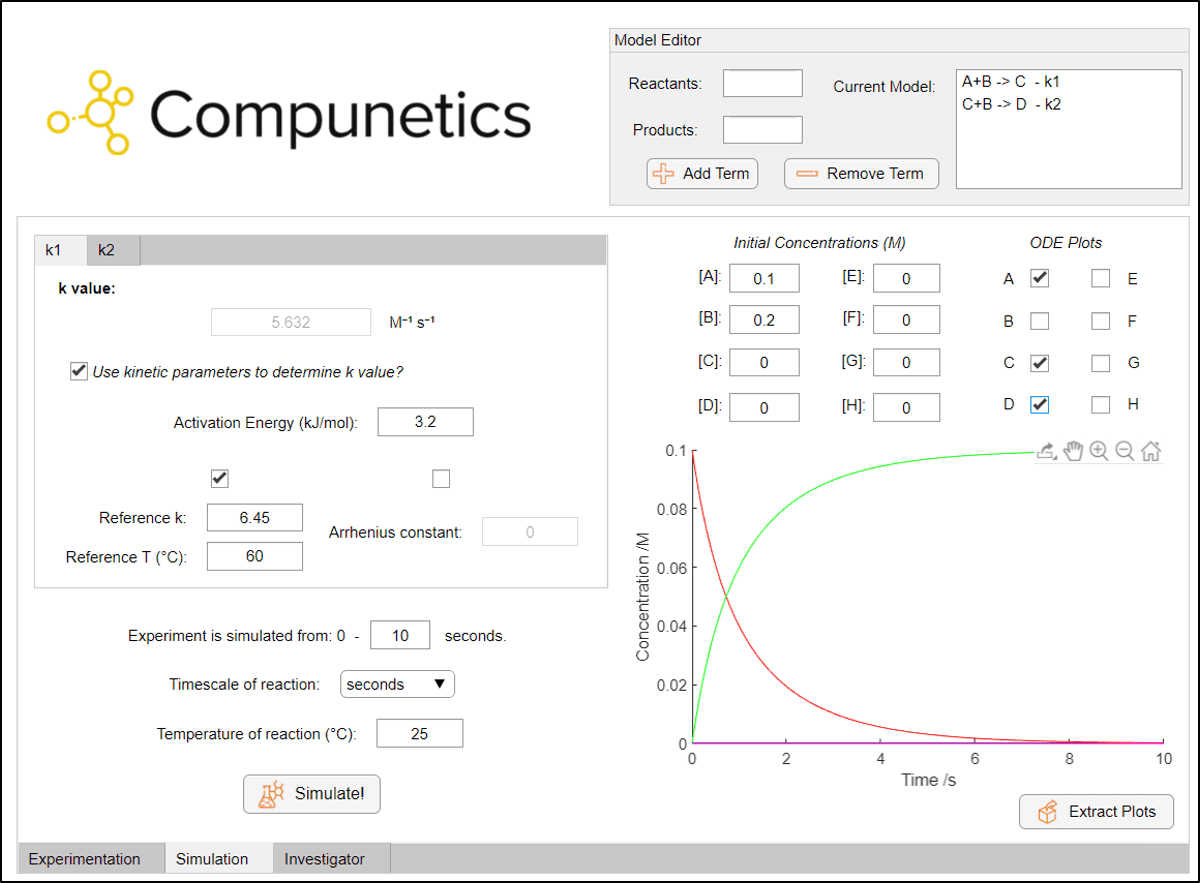
As a thought experiment, we can consider the degradation of acetic anhydride to be negligible - we will assume that all acetic anhydride is consumed from the reaction forming paracetamol and the reaction forming diacetamate. Using known kinetic parameters for this process from the literature[1] and the Simulation Toolbox in Compunetics, we can quickly and easily observe how our reaction is likely to proceed at different temperatures and reaction concentrations. Firstly, we input our reaction model and parameters into Compunetics:
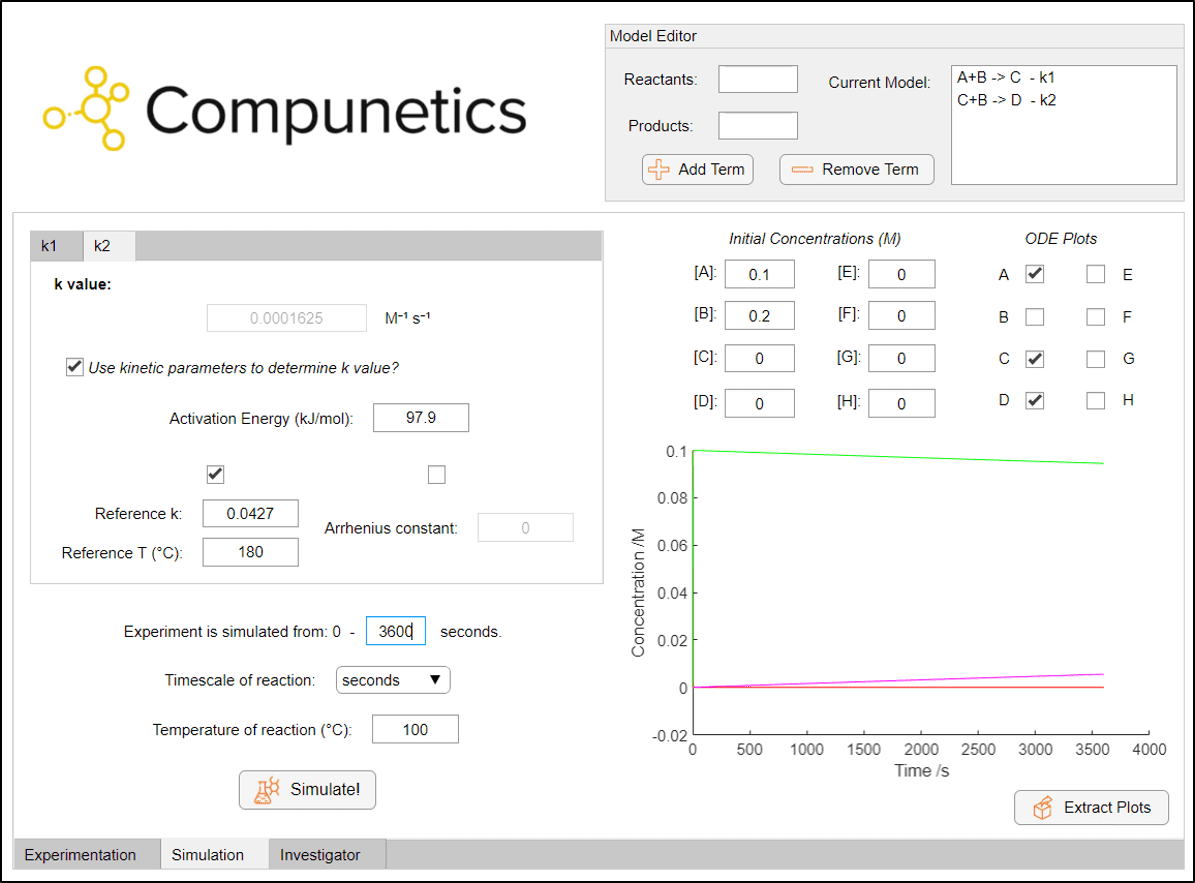
It can be noted instantly upon simulating reactions that, without the presence of the acetic anhydride degradation term, the disappearance of the 4-aminophenol starting material is very fast. Full conversion to paracetamol is predicted in less than 10 seconds at room temperature with a 0.1 M solution of 4-aminophenol. This is an interesting observation, as the preparation of paracetamol is often conducted at reflux for some time.[2] However, even running the reaction at a reflux temperature of 100 °C with these same initial concentrations doesn't result in a significant amount of diacetamate formation after one hour, with a yield of around 5 %.
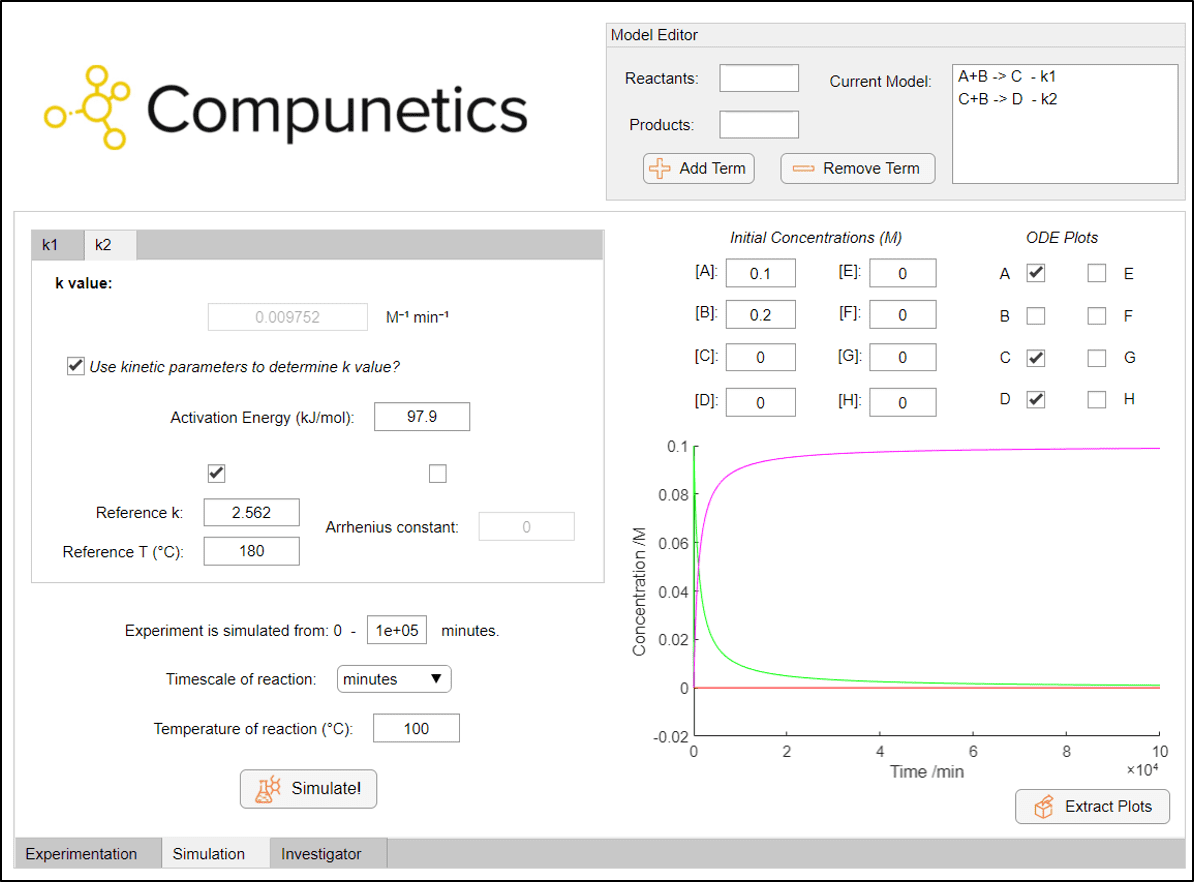
As can be seen, conducting the reaction at room temperature is likely to not form any diacetamate in measurable quantities. But to take this to the extreme case, how long would we need to reflux at 100 °C to obtain a 100 % yield of diacetamate (assuming no species degradation or solvent loss etc.)? Well, we can use Compunetics to quickly change the experimental timeframe to find out!
From adjusting the experimental timeframe, it's shown that it actually takes around 1 x 105 minutes (or approximately 70 days) to achieve full conversion of paracetamol to the byproduct diacetamate when running under constant reflux. This is made a lot easier using the Simulation Toolbox rather than running this in the lab to figure it out! So, the next time that you're running a paracetamol synthesis, rest assured that you probably won't form any over-reacted byproduct.
- Taylor, C. J. et al., Rapid Automated Determination of Reaction Models and Kinetic Parameters, Chemical Engineering Journal, 2020, 127017.
- Organic experiments, One-Day Practical Course for Year 12 Students, School of Chemistry, University of Leeds.