007. Reaction Simulation for Side-Product Prediction I
By Connor Taylor on November 13, 2020
One of the major benefits of conducting kinetic analysis is that a reaction can be simulated at different starting concentrations and temperatures, providing that the kinetic parameters are known. This allows the chemist to predict with confidence how the reaction should proceed upon changing these variables, rather than running further laboratory experiments which would cost significantly more time and resources to conduct. Typically, these simulations are performed by a physical-organic chemist with expertise in coding and mathematics, but with Compunetics any bench scientist can run them easily.
In the chemical process to synthesise paracetamol from 4-aminophenol and acetic anhydride, it is common to run the reaction in a suspension in water, although rarely does the experimenter achieve greater than a 70 % yield of paracetamol.[1] This could be for a number of reasons, such as formation of the diacetamate byproduct in Step Two, or less commonly considered is the degradation of the acetic anhydride to acetic acid.
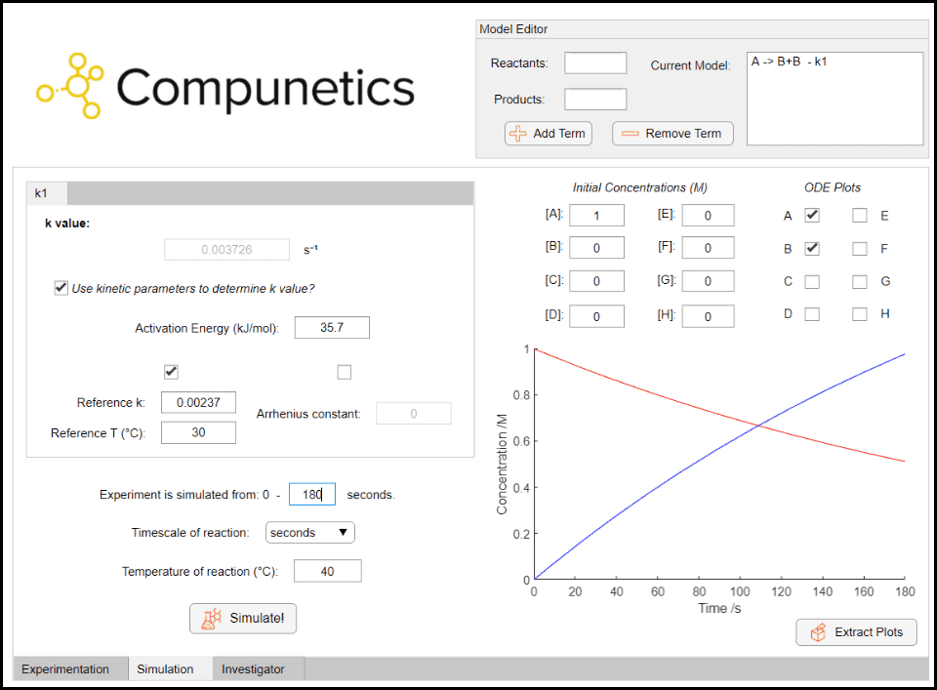
In this instance we would like to consider the significance of this degradation of our reagent, using known kinetic parameters and the Simulation Toolbox in Compunetics. We can use a pseudo-first order model shown below, as we know that the solvent (water) is in a large excess, and the kinetic parameters reported in the literature[2] to simulate the hydrolysis of acetic anhydride to unreactive acetic acid. The model and parameters are inputted into Compunetics, then the reaction can be quickly and easily simulated at different reaction conditions.
By quickly scanning different reaction conditions, it can be noted that the hydrolysis of acetic anhydride is very fast even at mild temperatures. If a 1 M concentration of acetic anhydride is present in water, after just 180 seconds at 40 °C, the concentration of this active reagent drops by almost 50 %. This can have a large affect on the yield of the desired paracetamol product but is seldom reported when considering this process. This information can serve as both practical quantitative information in the large-scale synthesis of paracetamol, but also in teaching kinetic analysis in the classroom as this can provide good insight into why desired yields are not obtained. These simulations in Compunetics, from the inputs of the kinetic parameters to the simulations and understanding of the chemistry was conducted in less than 5 minutes by an organic chemist with no prior kinetic expertise.
1: Comprehensive Organic Chemistry Experiments for the Laboratory Classroom, The Royal Society of Chemistry (2017).
2: Assirelli, M. et al., Reactor Kinetics Studies via Process Raman Spectroscopy, Multivariate Chemometrics, and Kinetics Modeling, Organic Process Research & Development, 2011, 15, 610-621.