004. Exploring and Quantifying the Caesium Effect I
By Thomas Dent on August 01, 2020
Caesium bases are routinely used in several industrially relevant palladium-catalysed coupling reactions such as Suzuki-Miyaura couplings and Buchwald-Hartwig aminations, and regularly excel as the optimal base in the literature. The mechanism by which caesium bases can improve yields and rates is not well understood, and this so called 'caesium effect' has anecdotally been attributed to factors such as increased solubility or the polarizability of the cation.
This report quantitatively screens several bases in the Buchwald-Hartwig amination of 4-iodoanisole, SM, and 4-methylaniline, An, to form the highest yield of the desired product, P, in both dimethylformamide (DMF) and toluene:
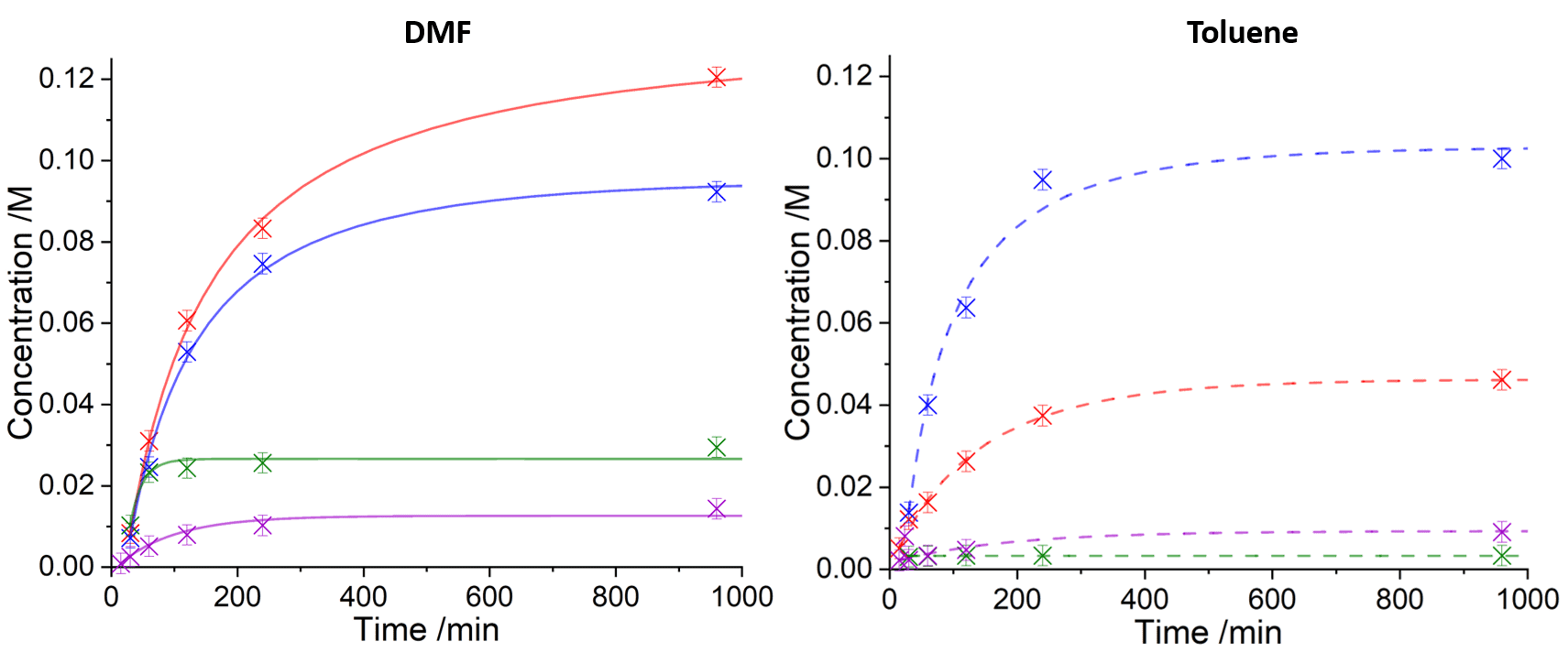
In this study, the effects of caesium carbonate (Cs2CO3) and caesium phosphate (Cs3PO4) on the yield of the product were explored. Benchmark reactions utilising potassium carbonate (K2CO3) and potassium phosphate (K3PO4) were also conducted. The kinetic profiles were monitored using NMR, and modelled using observed rate constants (with a corresponding palladium degradation pathway) easily with Compunetics. Here, the degraded palladium species is denoted as dPd.
DMF
| Cs2CO3 | Cs3PO4 | K2CO3 | K3PO4 | |
|---|---|---|---|---|
| k1 /M2 s-1 | 0.1076 | 0.1009 | 0.1037 | 0.0159 |
| k2 /s-1 | 9.50E-06 | 5.72E-05 | 0.0008 | 0.0002 |
Toluene
| Cs2CO3 | Cs3PO4 | K2CO3 | K3PO4 | |
|---|---|---|---|---|
| k1 /M2 s-1 | 0.0386 | 0.1450 | 0.1001 | 0.0049 |
| k2 /s-1 | 9.21E-05 | 7.92E-05 | 0.0626 | 9.04E-05 |
As shown from the observed kinetics, when all experimental conditions are constant apart from the base, the caesium bases typically outperform the potassium bases when measuring conversion to product. For example, in DMF, the use of Cs2CO3 results in a yield of the product 4x greater than when using K2CO3, and the use of Cs3PO4 results in a yield 6x greater than when using K3PO4. As only observed rate constants were measured, deep mechanistic insights cannot be obtained regarding the effects of the caesium cation, but the rate constants suggest that the palladium degradation pathways may be inhibited more when using its respective bases in reactions.
Depending on the suitability of these solvents (DMF and toluene) in the scale-up of this process, decisions regarding solvent selection can then be made.[2] This early developmental stage, however, has benefited from the kinetic fittings by Compunetics, by providing easy-to-use quantitative analysis of the chemical system which can be utilised in further process development.
- Dent, T.J., Controlling and exploiting the caesium effect in palladium catalysed coupling reactions, Thesis, 2019.
- Alder, C.M. et al, Updating and further expanding GSK's solvent sustainability guide, Green Chem., 2016, 18, 3879-3890.