003. Simulation and Optimisation of a Fine Chemical Steroid
By Jamie Manson on July 21, 2020
A major benefit to conducting kinetic analysis is that if the model and kinetic parameters are known, simulated experiments can be conducted at different starting material concentrations and temperatures. This has major implications on the pharmaceutical and fine chemical industries, as a scientist can accurately predict how a reaction will proceed based on this information, without the need to carry out the reaction themselves. More importantly, this also allows a chemist to simulate and identify optimum reaction conditions for process development. Compunetics provides the tools for bio/chemists so that they can easily simulate these reactions and optimise their processes in silico, without the need to even enter the lab.
When considering the transformation of the steroid derivative, SM, with the common fluorinating agent, Deoxofluor, there are two products from the reaction: the desired product, P1, and the unwanted by-product, P2:
The kinetic model and parameters can then be inputted directly and intuitively into the Compunetics Simulation Toolbox. This model contains the two predicted reactions, k1 and k2, but also features a first-order degradation pathway whereby Deoxofluor forms degraded and inactive impurities, with the rate constant k3. This could lead to complications during conventional process development, but the identification of this pathway and the derived kinetics allows the scientist to predict how this will affect further experimentation.
| k90 °C /M-1 min-1 or min-1 | Ea /kJ mol-1 | |
|---|---|---|
| k1 | ||
| k2 | ||
| k3 |
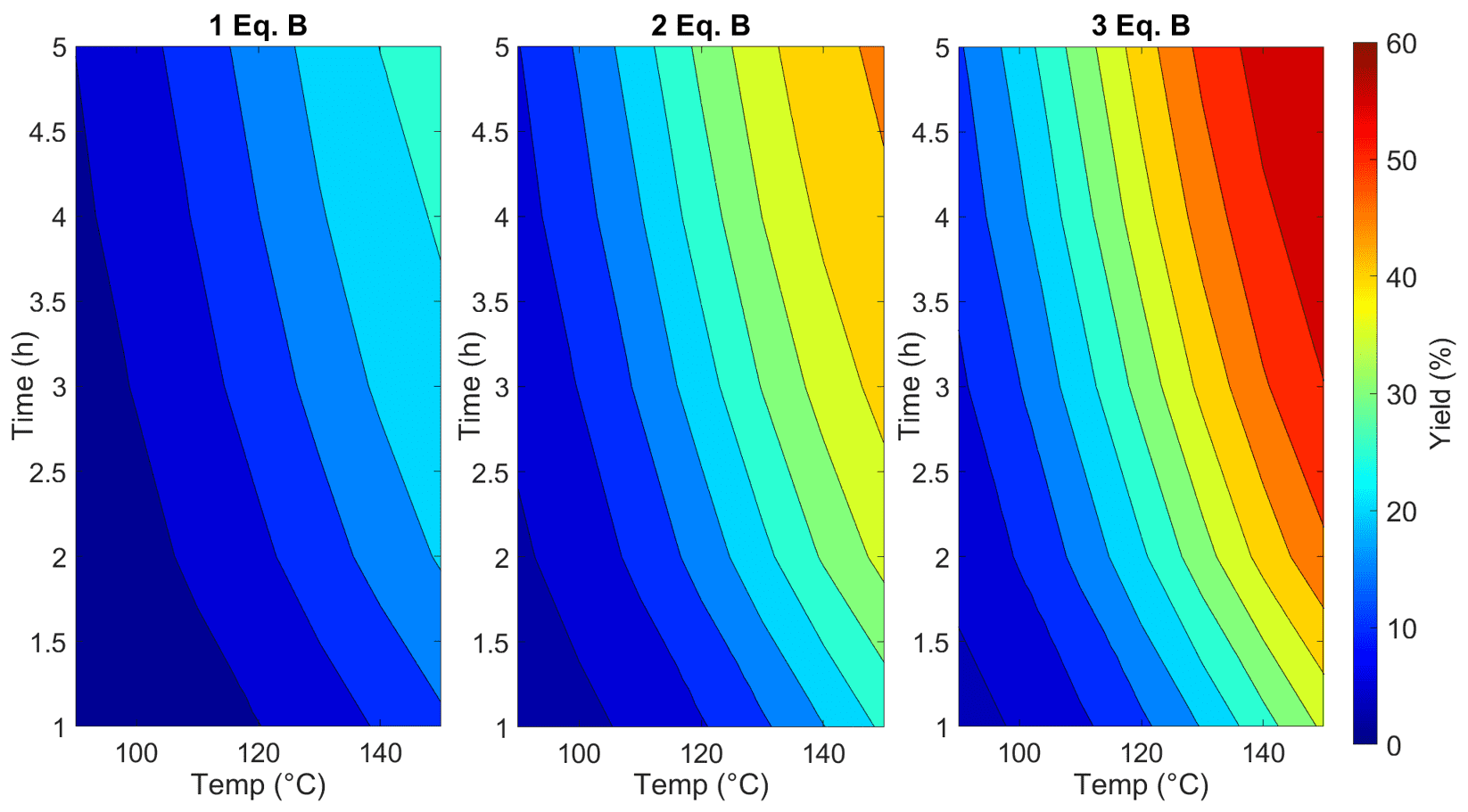
There are then several ways to optimise this reaction in silico, but one of the simplest methods is to plot a response surface in a graphing software such as Origin, or alternatively on a MATLAB/Python platform. The upper and lower bounds must initially be identified by the researcher for each of the experimental variables, which for this process were: temperature (90 - 150 °C), reaction time (1 - 5 hours) and Deoxofluor equivalents (1 - 3), where the initial concentration of SM is 1 M. Equidistant experimental simulations with differing factors can then be conducted instantly in Compunetics, exporting the simulated result of each experiment in order to build a response surface. This response surface (which in this case comprises 30 simulated experiments) allows the chemist to identify regions of optimal experimental design space, as well as the corresponding yields to be expected from conducting a real experiment at these conditions.
It has been shown that process development and in silico optimisation can be achieved easily by utilising the Simulation Toolbox within Compunetics, followed by building a response surface in common graphing software. Experiments that would otherwise take 5 hours to run can be simulated instantly, saving both time and material costs, in order to identify regions of optimal experimental design space. It was found in this reaction system that higher temperatures, reaction times and Deoxofluor equivalents drive the reaction forward without significant unwanted by-product formation, achieving optimised yields of the desired product (P1) at around 60%.
Utilising in silico optimisation for process development and research is a key tool that is unlocked via the use of Compunetics, which is becoming ever more important especially as experimental time remains limited for many researchers due to COVID-19.
1. Negi, Devender S. et al., Organic Process Research & Development, 2008, 12, 345-348.