002. Estimating Reaction Completion
By Connor Taylor on July 05, 2020
One major benefit to conducting kinetic analysis is that when the kinetic parameters of a reaction are known, the reaction can be simulated at different concentrations and temperatures. This allows the chemist to predict with confidence how the reaction proceeds upon changing these variables, which can save time and money that would otherwise be used in further conducting exploratory experiments. When considering the synthesis of the building block, Prod, from 4-chlorobenzoic acid, SM, we can utilise known kinetic information to influence how to conduct this process:
As the intermediate, Int, is the only downstream product from the first reaction, it becomes possible to conduct the second step without isolation of Int, by sequentially adding the necessary reagents to the same flask. However, it is important that there is no SM remaining upon addition of the Buchwald-Hartwig amination reagents for the second step, as this may result in unwanted side-products. To ensure that we select reaction conditions whereby all SM is consumed in the first reaction, and hence maximise the potential yield of Prod, we can simulate this reaction easily using Compunetics and reported kinetic parameters.
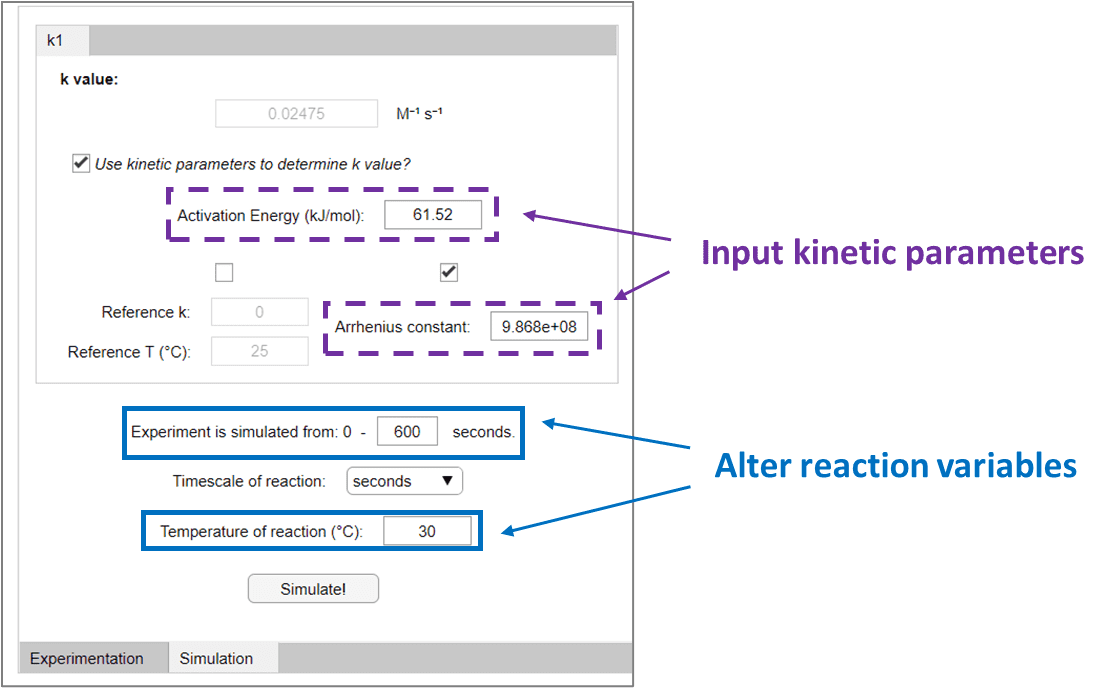
Compunetics is built specifically for intuitive use for the typical bench scientist, without the need for coding, mathematical or kinetic expertise. The reaction of SM and CH3I to form Int, can be inputted into Compunetics in more simplistic terms as A + B → C. The known kinetic parameters can then also be entered, as well as the desired reaction variables. These values can be changed seamlessly and independently of each other, to instantly simulate how the reaction should proceed.
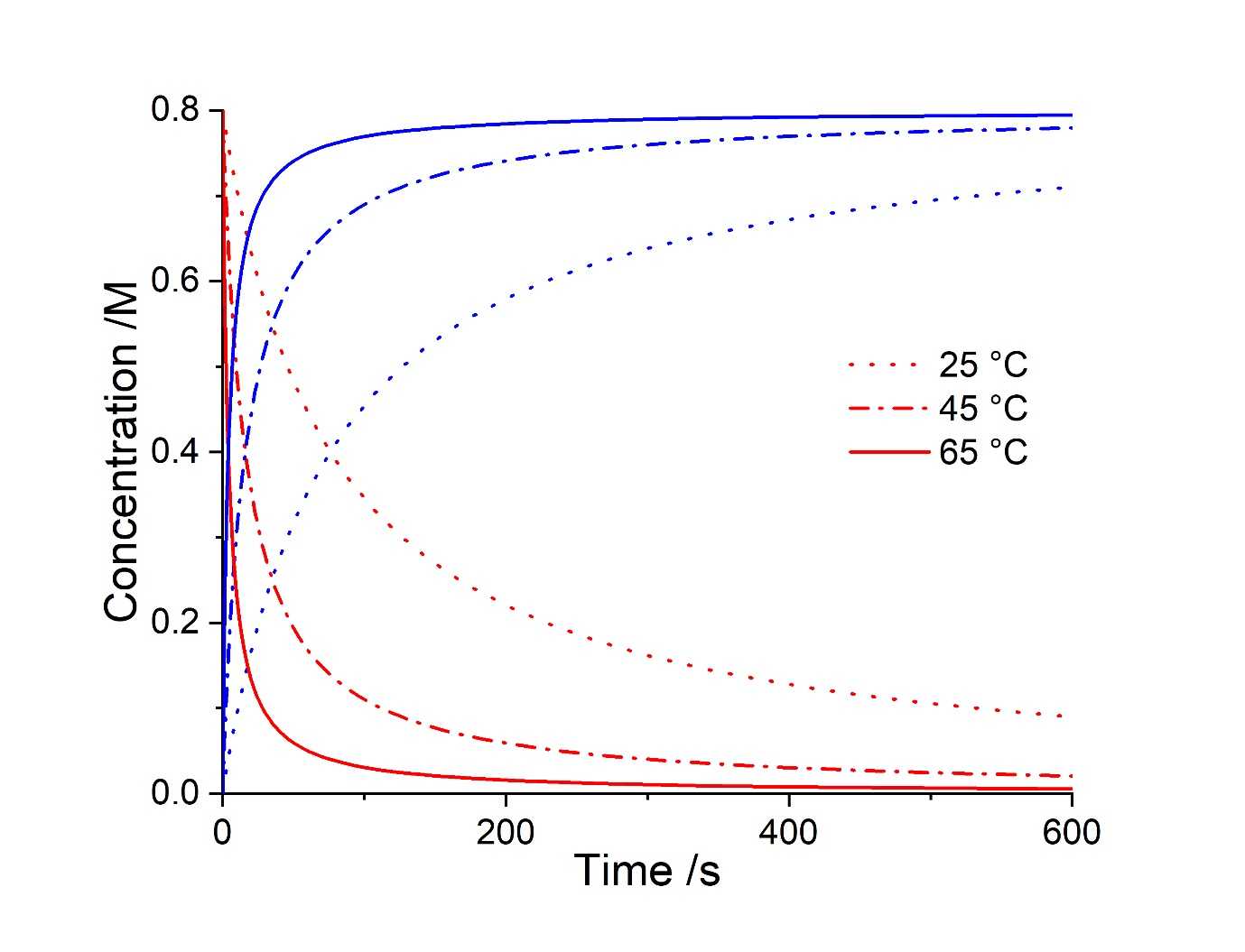
In this hypothetical example, it is 4 pm on a Friday and you would like to join your colleagues at the bar as a fellow research member has just passed their PhD viva - you therefore need to be fast in setting up your reaction. You want the first reaction to be complete, and hence all SM consumed, within 10 minutes, so that you can add your Buchwald-Hartwig amination reagents and go to congratulate your new doctor friend, as the experiment forms the Prod species over the weekend. Therefore, you would like to alter the temperature of your simulated reaction in Compunetics (0.8 M SM, 0.8 M CH3I, 0.8 M TMGN) in order to identify the correct temperature at which to run your experiment. This temperature can be altered to immediately produce updated simulated kinetic plots, so that the desired temperature can be determined.
It is shown that by running this experiment at 65 °C, almost all of the SM is consumed within the first 10 minutes of the reaction, at which point it is appropriate to add the reagents for the second step. This brief example has shown how Compunetics can be used to simulate exploratory reactions for a reaction system, when kinetic parameters are known. These simulations reflect real-world experimentation and can save unnecessary money- and time-expenditure, as all computation was conducted in less than 2 minutes (with no further material consumption). Compunetics allows any chemist to easily utilise reaction simulation as part of their synthetic toolkit, which until now has been otherwise unavailable and reserved for coders, mathematical experts and kineticists.
- Gholamipour-Shirazi, A. and Rolando, C., Organic Process Research & Development, 2012, 16, 5, 811-818.
- Reddy, Ch. Venkat et al., Journal of Organic Chemistry, 2008, 73, 8, 3047-3062.