001. Quantifying Catalytic Activity
By Connor Taylor on June 16, 2020
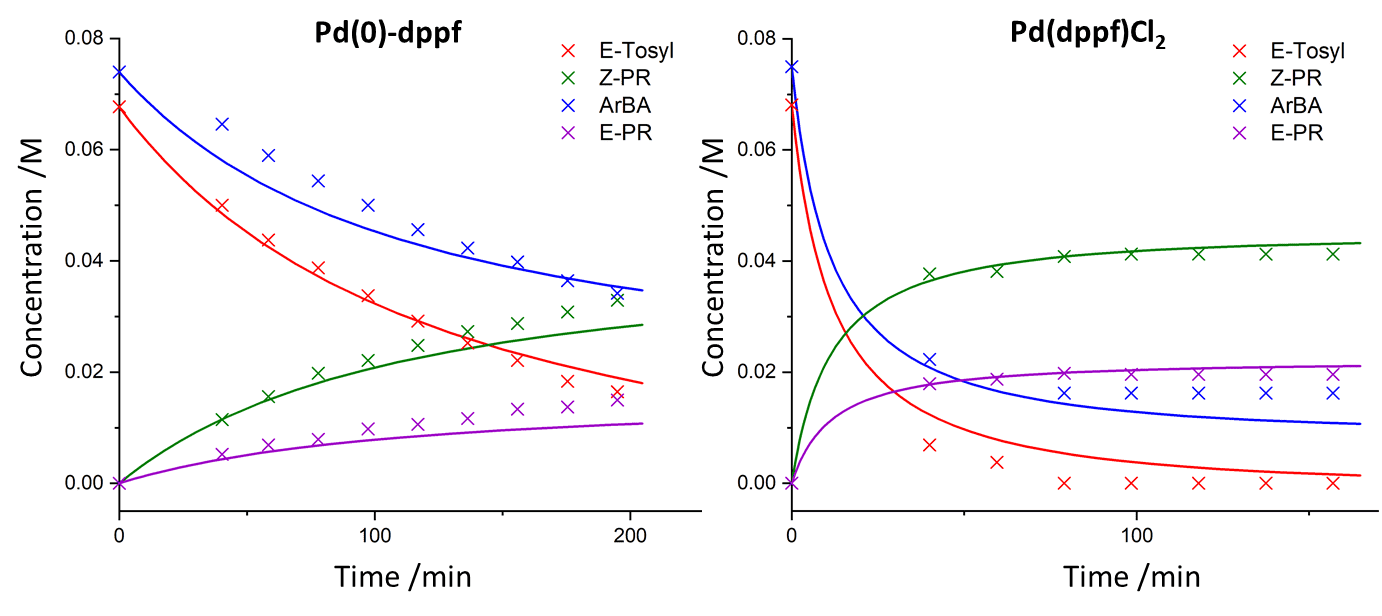
Recently, Christensen et al. reported the use of an automated kinetic profiling methodology to explore and optimise the palladium-catalysed Suzuki-Miyaura reaction to generate a cinnamate derivative for a drug discovery program. [1] This reaction features the coupling of the vinyl tosylate, E-Tosyl, with the aryl boronic acid, ArBA, to form the desired product, E-PR, as well as an identified side-product, Z-PR. As part of the reaction scope, two palladium catalysts were utilised: Pd(0)-dppf and Pd(dppf)Cl2.
Using the data from this report by Christensen et al., quantitative kinetic analysis for the two catalysts was performed using Compunetics. The empirical third order rate laws for the two major reactions, k1 and k2, were fitted for both sets of kinetic data - one set of data for each palladium catalyst. Rather quickly it was noted that the disappearance of the starting material, E-Tosyl, was greater than the disappearance of the boronic acid, ArBA. Therefore, a degradation pathway to an unknown side product (SP) was assumed and inputted as a third reaction, k3. This value for k3 was first fitted in the Pd(0)-dppf reaction, then utilising the Compunetics options allowed this k3 value to remain constant for fitting of the data using Pd(dppf)Cl2, as the degradation was assumed to be independent of catalyst. These observed rate constants were recorded and the fit to the experimental data was plotted.
| Pd(0)-dppf | Pd(dppf)Cl2 | |
|---|---|---|
Quantitative kinetic analysis has shown that utilising the palladium catalyst Pd(dppf)Cl2 for this Suzuki-Miyaura reaction is much faster than the considered Pd(0)-dppf catalyst. Observed reactions rates exceed a ten-fold (10x) increase when Pd(dppf)Cl2 is employed, meaning that this catalyst is likely to be optimal for a higher throughput of desired product. Further in silico studies can confirm this upon decisions to scale-up the reaction during process development. All kinetic data/reaction model input and kinetic parameter evaluations were performed in less than 2 minutes using Compunetics, showing that rapid, accurate kinetic analysis can be performed easily for catalytic processes with real-world practical applications.
- Christensen, M. et al., Development of an automated kinetic profiling system with online HPLC for reaction optimization. Reaction Chemistry & Engineering, 2019. 4(9), 1555-1558. doi: 10.1039/C9RE00086K